Catalytic asymmetric synthesis of 3,2'-pyrrolinyl spirooxindoles via conjugate addition/Schmidt-type rearrangement of vinyl azides and (E)-alkenyloxindoles. - Abstract - Europe PMC

Divergent synthesis of chiral cyclic azides via asymmetric cycloaddition reactions of vinyl azides | Nature Communications

Metal‐Free Electrochemical Coupling of Vinyl Azides: Synthesis of Phenanthridines and β‐Ketosulfones - Li - 2020 - European Journal of Organic Chemistry - Wiley Online Library
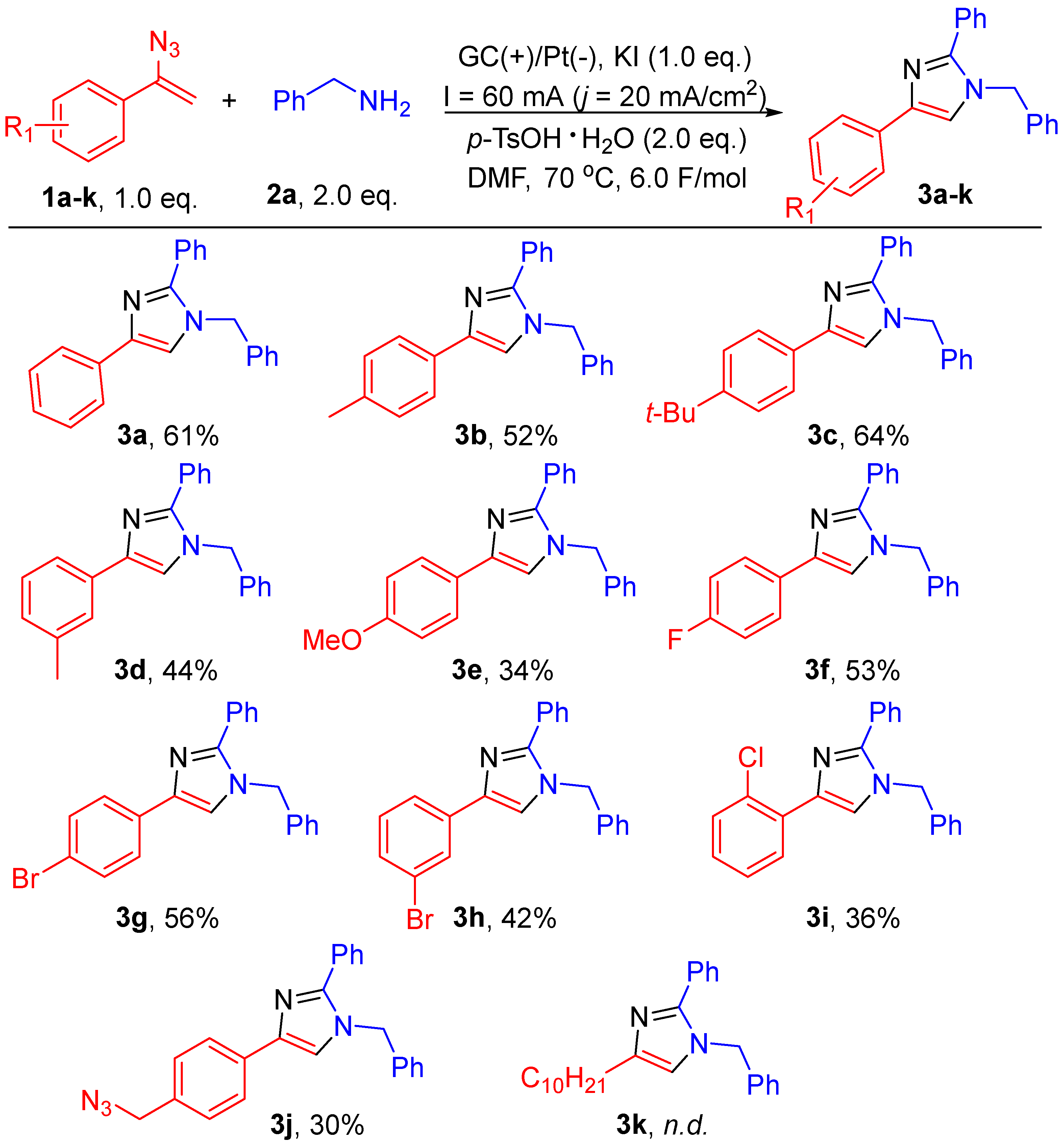
Molecules | Free Full-Text | Electrochemically Induced Synthesis of Imidazoles from Vinyl Azides and Benzyl Amines

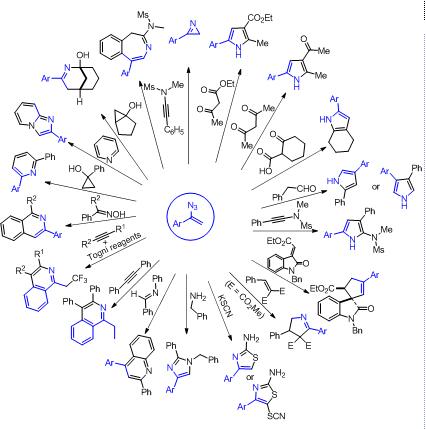
Application of Vinyl Azides in Chemical Synthesis: A Recent Update | The Journal of Organic Chemistry

Application of Vinyl Azides in Chemical Synthesis: A Recent Update | The Journal of Organic Chemistry
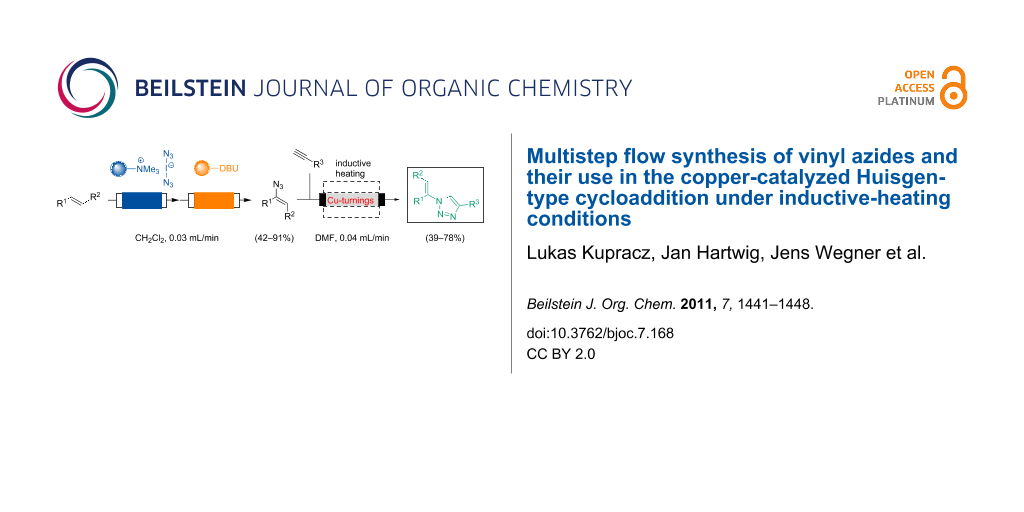
BJOC - Multistep flow synthesis of vinyl azides and their use in the copper-catalyzed Huisgen-type cycloaddition under inductive-heating conditions
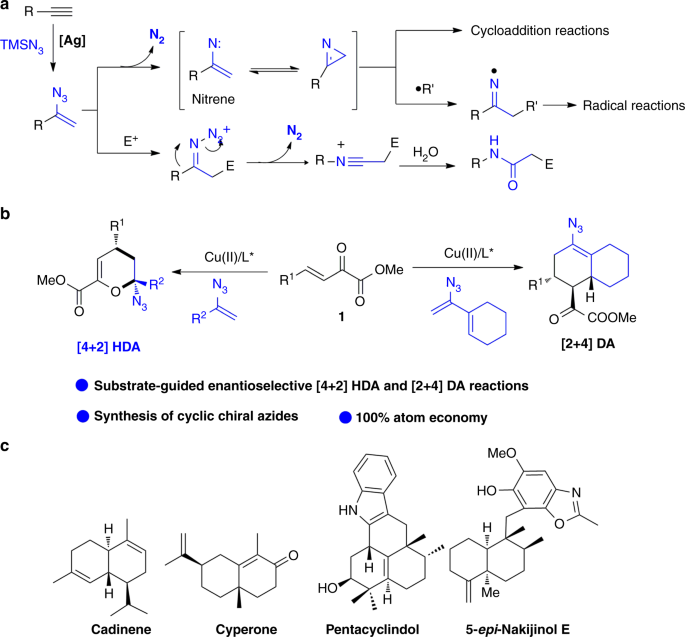
Divergent synthesis of chiral cyclic azides via asymmetric cycloaddition reactions of vinyl azides | Nature Communications
![Formal [4+2]‐Annulation of Vinyl Azides with N‐Unsaturated Aldimines - Zhu - 2014 - Chemistry – An Asian Journal - Wiley Online Library Formal [4+2]‐Annulation of Vinyl Azides with N‐Unsaturated Aldimines - Zhu - 2014 - Chemistry – An Asian Journal - Wiley Online Library](https://onlinelibrary.wiley.com/cms/asset/0aa426d1-2d8f-4238-9373-2604080ae4b5/msch001.jpg)
Formal [4+2]‐Annulation of Vinyl Azides with N‐Unsaturated Aldimines - Zhu - 2014 - Chemistry – An Asian Journal - Wiley Online Library

Scheme 7: Formation of 2,5-dihydrooxazole 9 starting from vinyl azide... | Download Scientific Diagram

Ene-gamma-lactam synthesis via (3+2) vinyl azide-enolate cycloaddition: Enolate addition to vinyl triazolinone intermediates | Organic Chemistry | ChemRxiv | Cambridge Open Engage
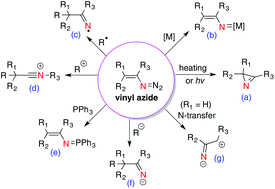
Reactivities of vinyl azides and their recent applications in nitrogen heterocycle synthesis - Organic & Biomolecular Chemistry (RSC Publishing)

Scheme 74. Cycloaddition Reactions of Vinyl Azides for the Synthesis of... | Download Scientific Diagram
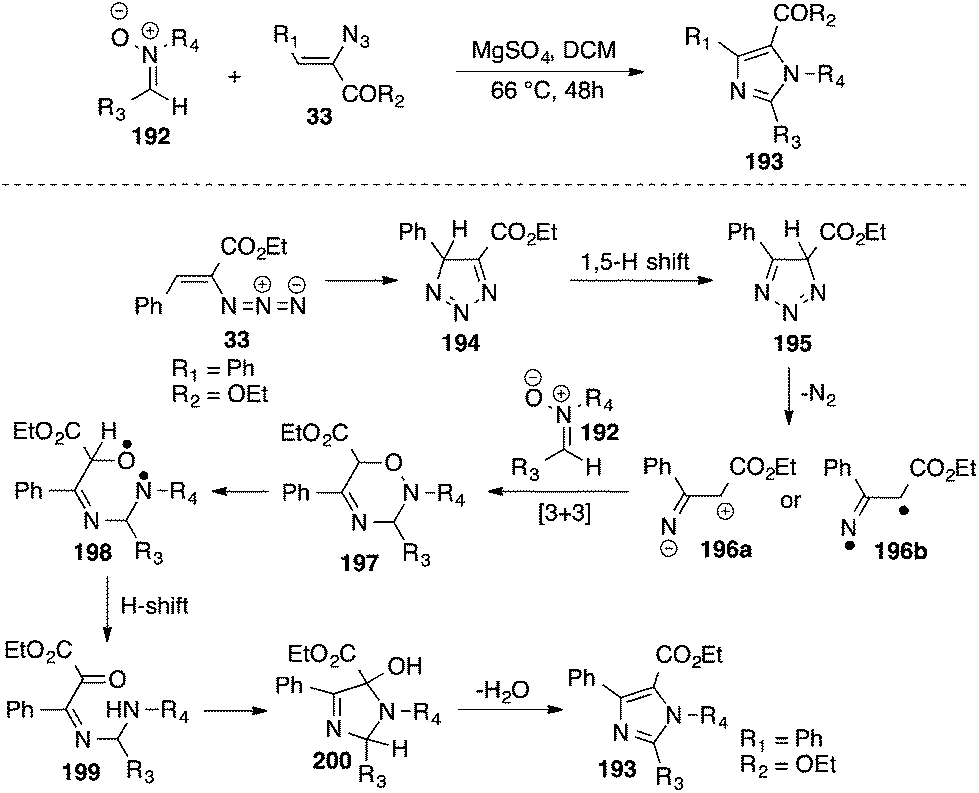
Reactivities of vinyl azides and their recent applications in nitrogen heterocycle synthesis - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C5OB00099H

Scandium (III)‐Catalyzed Cycloaddition of in situ Generated ortho‐Quinone Methides with Vinyl Azides: An Efficient Access to Substituted 4H‐Chromenes - Thirupathi - 2018 - Advanced Synthesis & Catalysis - Wiley Online Library
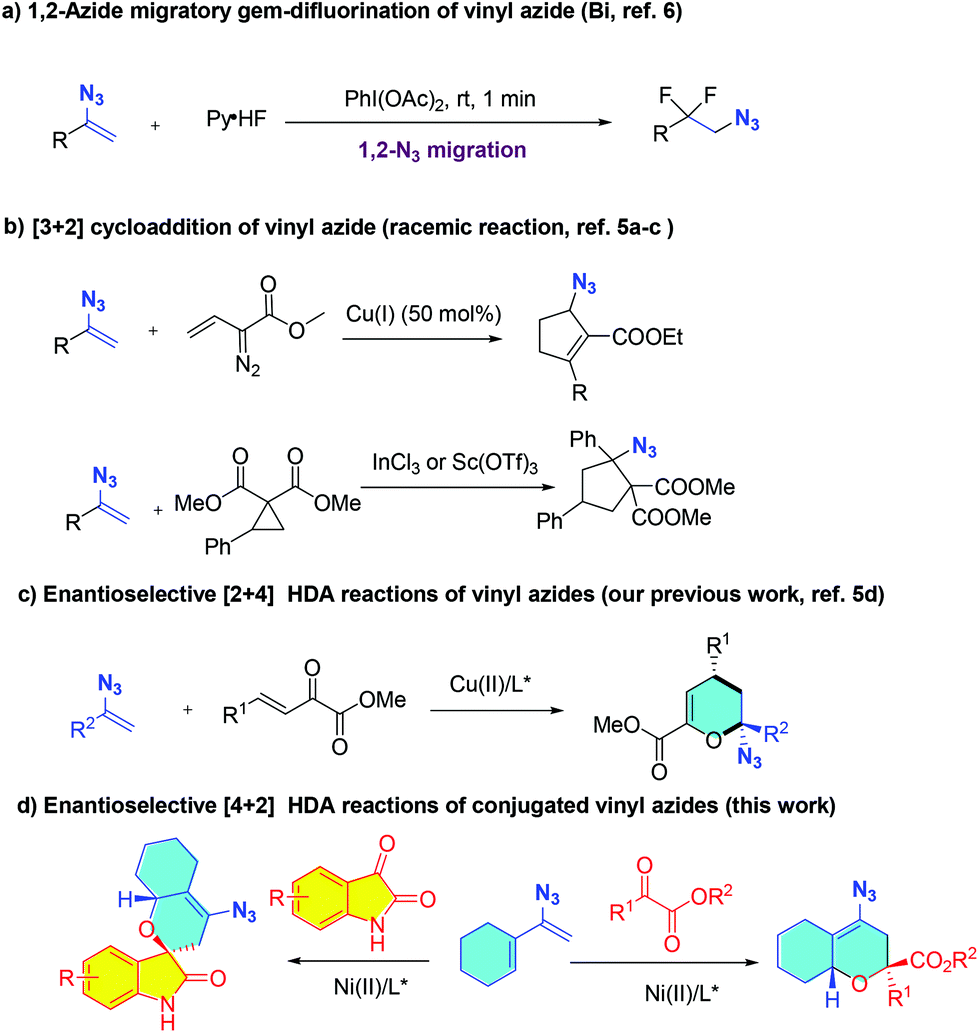
Ni-Catalyzed asymmetric hetero-Diels–Alder reactions of conjugated vinyl azides: synthesis of chiral azido polycycles - Organic Chemistry Frontiers (RSC Publishing) DOI:10.1039/D0QO01597K
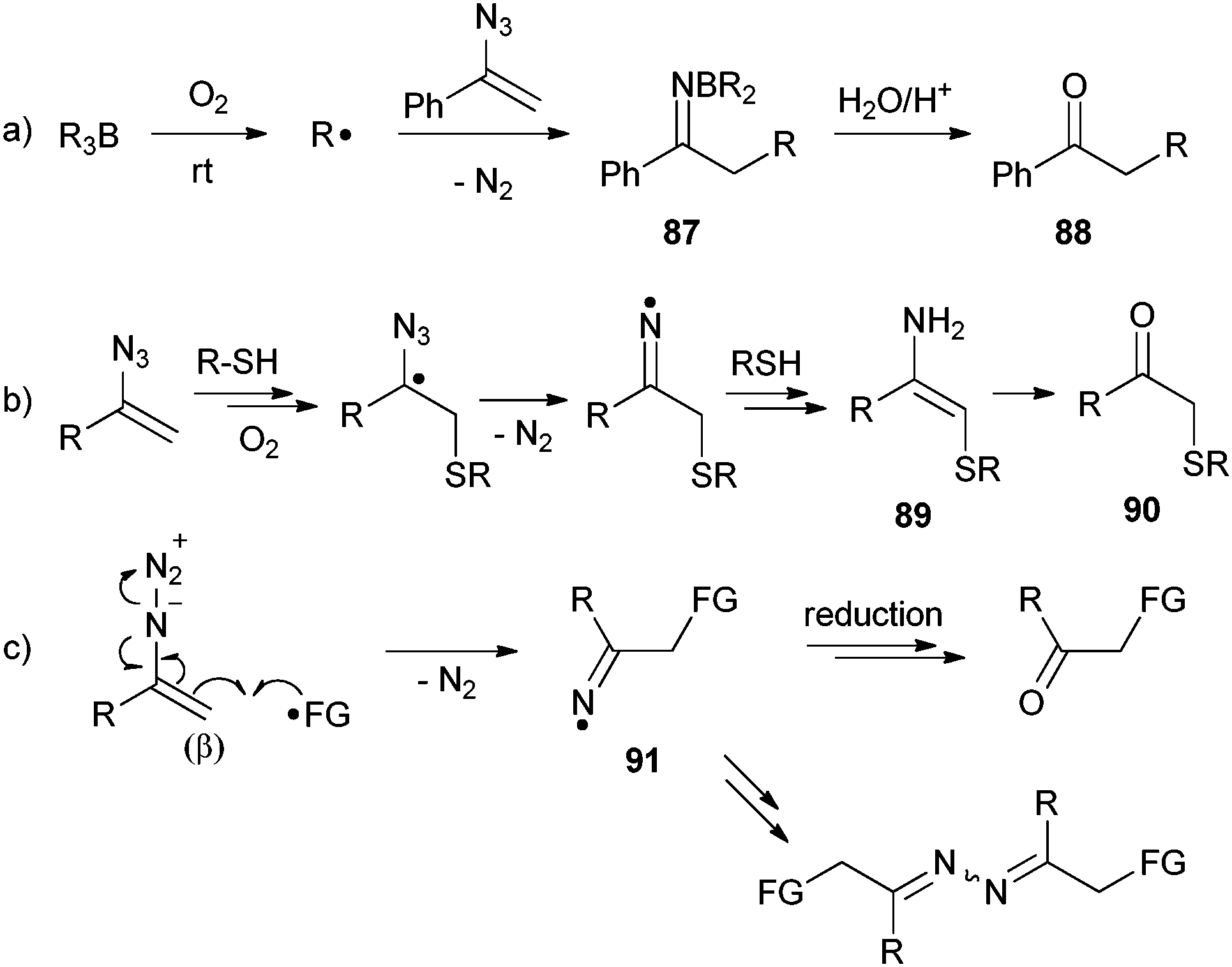
α-Substituted vinyl azides: an emerging functionalized alkene - Chemical Society Reviews (RSC Publishing) DOI:10.1039/C7CS00017K
![PDF] Radical Enamination of Vinyl Azides: Direct Synthesis of N-Unprotected Enamines. | Semantic Scholar PDF] Radical Enamination of Vinyl Azides: Direct Synthesis of N-Unprotected Enamines. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/ad4e62b83576af821a221a0a20bd8ad9ee8a9ff9/2-Table1-1.png)