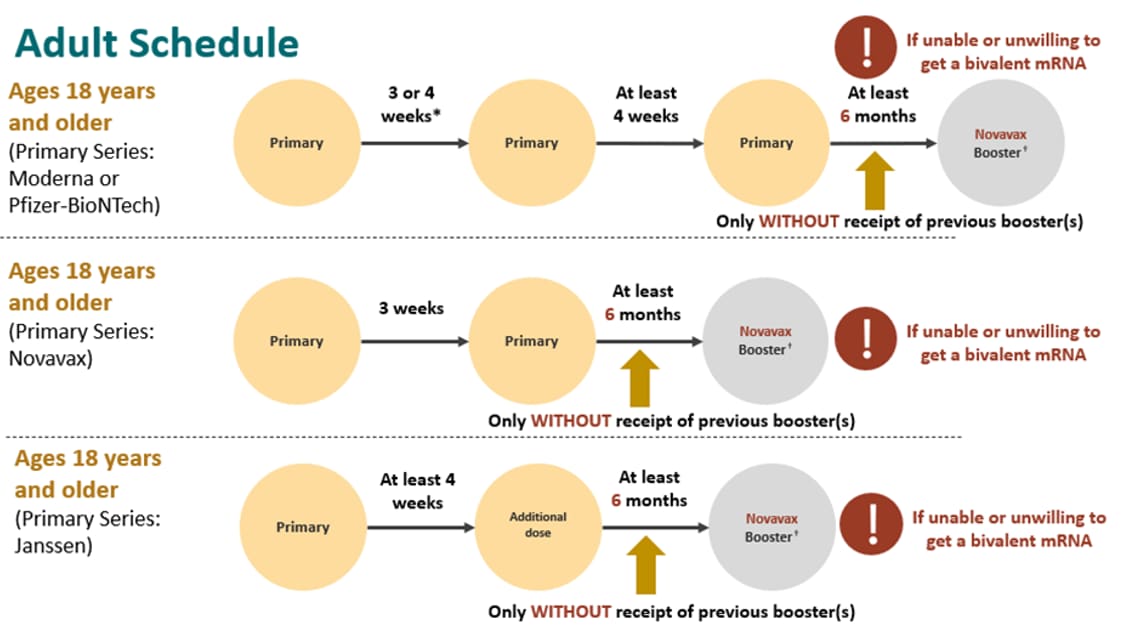
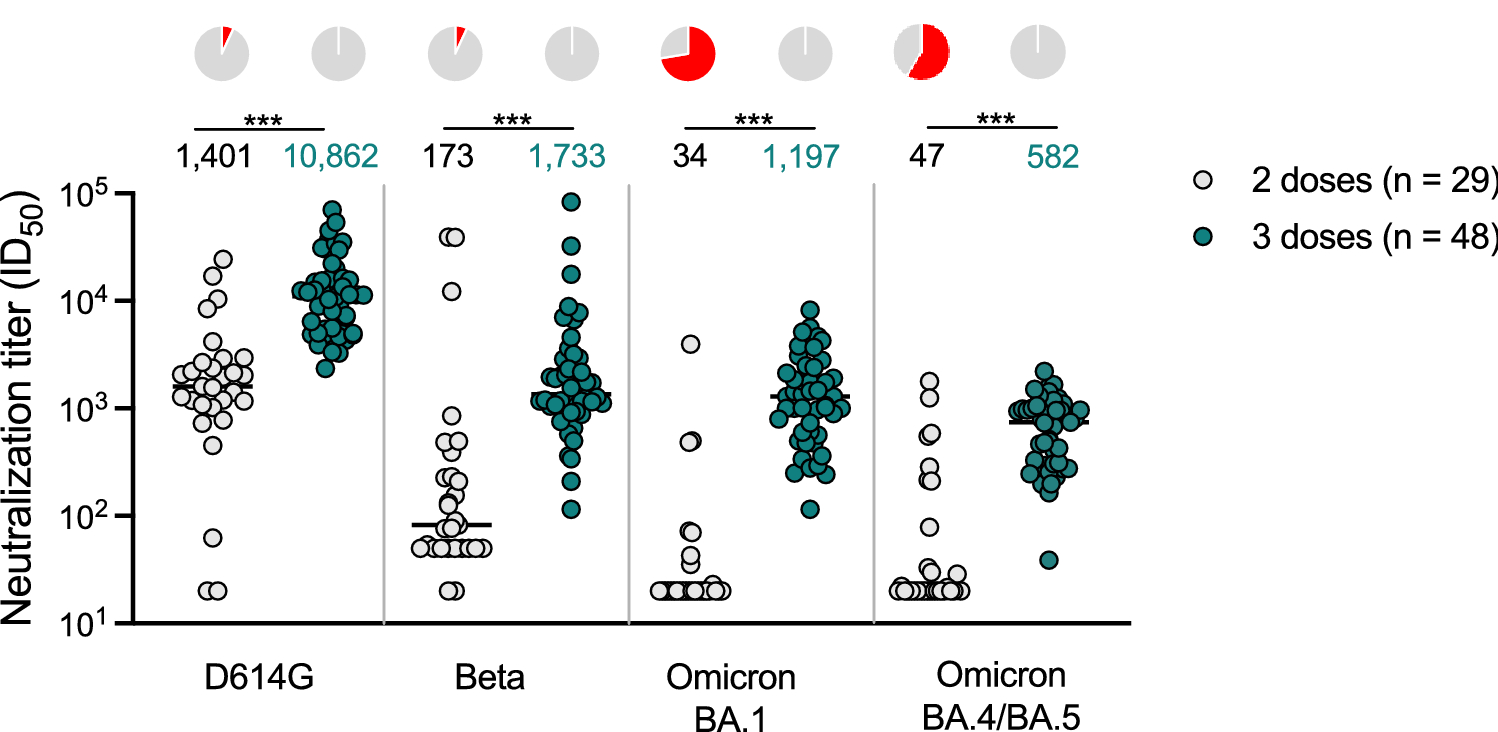
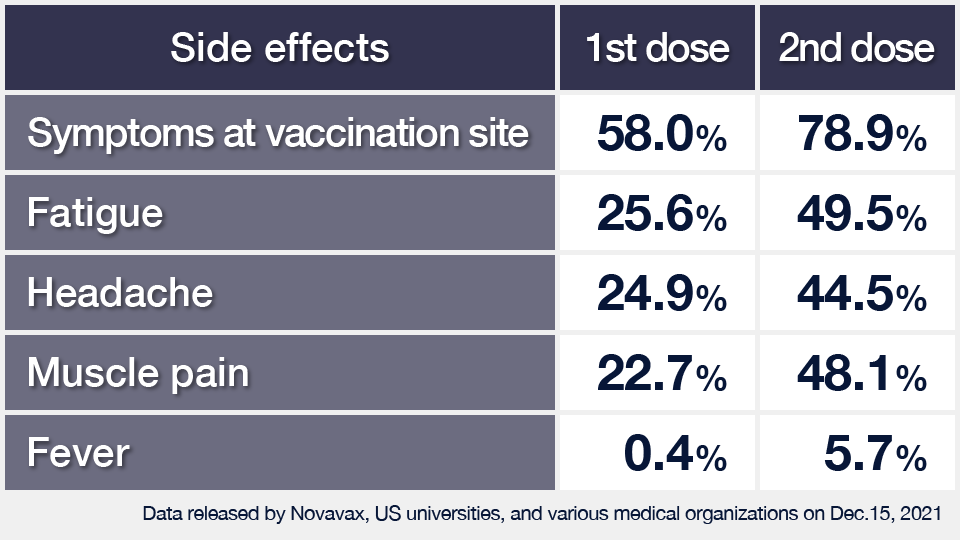
Novavax on Twitter: "A 3rd dose boost increased functional hACE2 inhibition responses against variants. Response vs Delta (B.1.617.2) variant was >6x higher than after the primary series, >10x for Beta (B.1.351) &

Novavax and J&J COVID-19 vaccines are effective, but the virus variant from South Africa poses problems
News - European Commission Grants Conditional Marketing Authorisation for Novavax's COVID-19 Vaccine Nuvaxovid - Paul-Ehrlich-Institut

Novavax's long-awaited COVID-19 vaccine authorizations offer an alternative to mRNA | Science | AAAS

News - Nuvaxovid (Novavax) – Protein-based COVID-19 Vaccine Available on a Limited Basis in Germany - Paul-Ehrlich-Institut
New research suggests booster dose of Novavax NVX-CoV2373 vaccine is effective against SARS-CoV-2 Omicron subvariants