
PDF) Reduction of imatinib dose and persistence of complete molecular response after p210 multipeptide vaccine in chronic myeloid leukaemia treated with dose escalation for acquired resistance | Michelina Santopietro - Academia.edu
Imatinib dose reduction in major molecular response of chronic myeloid leukemia: results from the German Chronic Myeloid Leukemia-Study IV | Haematologica

Imatinib dose escalation for chronic phase–chronic myelogenous leukaemia patients in primary suboptimal response to imatinib 400 mg daily standard therapy | Leukemia

Dasatinib vs. imatinib in patients with chronic myeloid leukemia in chronic phase (CML-CP) who have not achieved an optimal response to 3 months of imatinib therapy: the DASCERN randomized study | Leukemia

Predicting toxicity for patients with advanced Gastrointestinal Stromal Tumors (GIST) treated with imatinib mesylate : an EORTC/ISG/AGITG randomized trial. - ppt download

Low‐dose tyrosine kinase inhibitors before treatment discontinuation do not impair treatment‐free remission in chronic myeloid leukemia patients: Results of a retrospective study - Cayssials - 2020 - Cancer - Wiley Online Library

Therapeutic drug monitoring of imatinib in patients with gastrointestinal stromal tumours – Results from daily clinical practice - ScienceDirect

Switching to nilotinib versus imatinib dose escalation in patients with chronic myeloid leukaemia in chronic phase with suboptimal response to imatinib (LASOR): a randomised, open-label trial - The Lancet Haematology
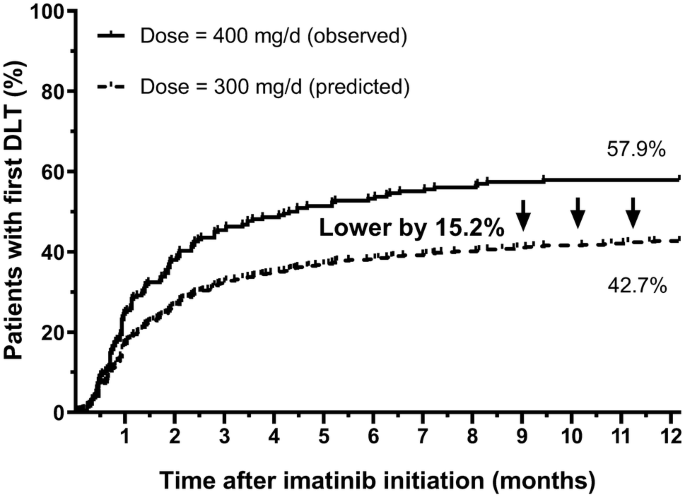
Comprehensive analyses of safety and efficacy toward individualizing imatinib dosage in patients with chronic myeloid leukemia | SpringerLink
![PDF] Imatinib dose escalation versus sunitinib as a second-line treatment against advanced gastrointestinal stromal tumors: A nationwide population-based cohort study. | Semantic Scholar PDF] Imatinib dose escalation versus sunitinib as a second-line treatment against advanced gastrointestinal stromal tumors: A nationwide population-based cohort study. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/16cb8478b80a5ef34b1c1ac8f4115a98c8fe80ff/5-Figure2-1.png)













