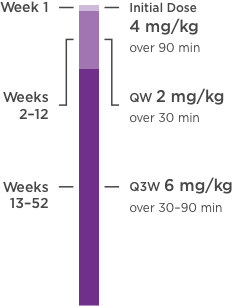
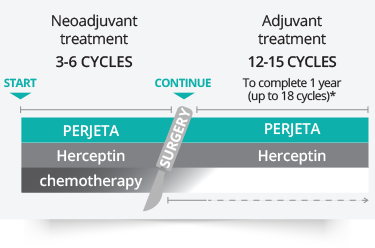
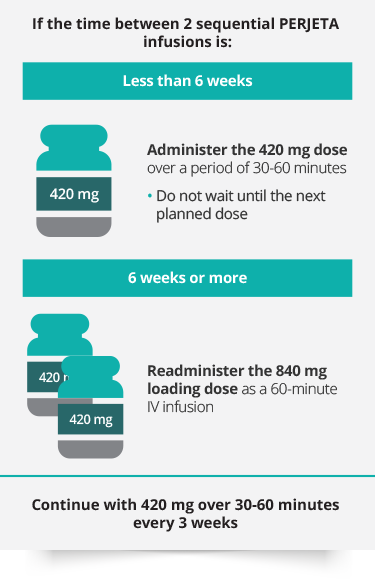
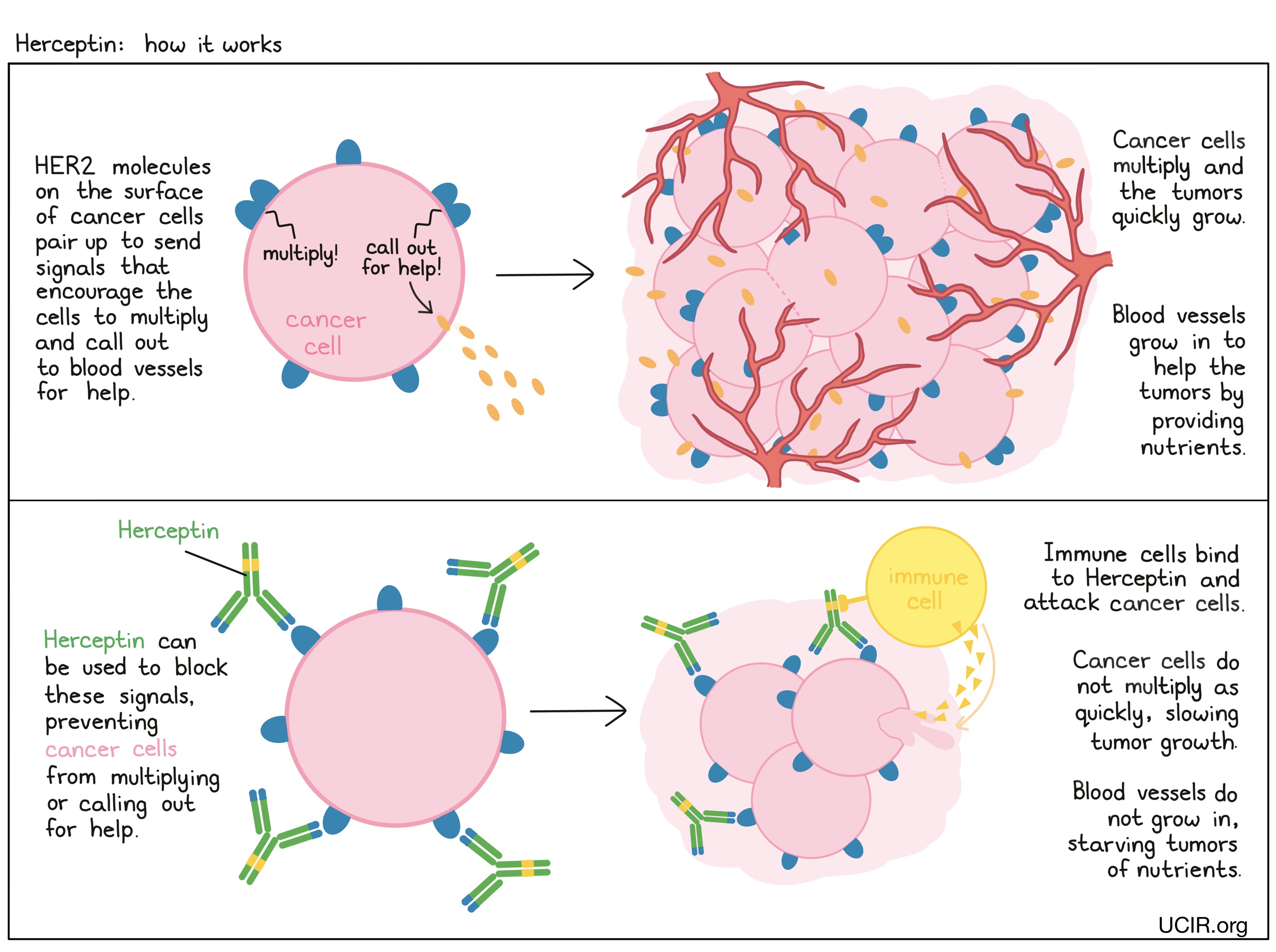
Development of a Subcutaneous Fixed‐Dose Combination of Pertuzumab and Trastuzumab: Results From the Phase Ib Dose‐Finding Study - Kirschbrown - 2019 - The Journal of Clinical Pharmacology - Wiley Online Library
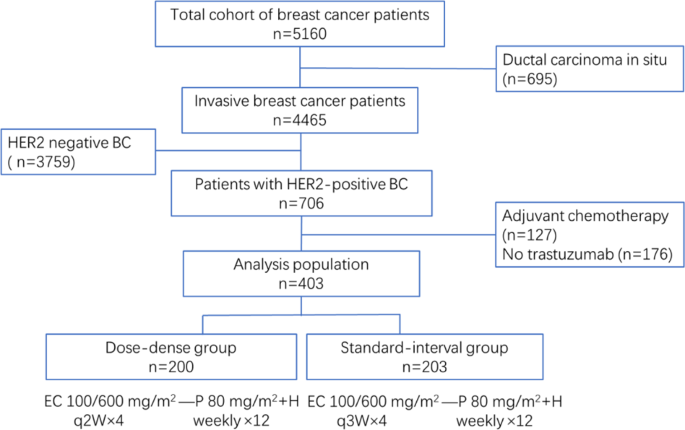
Impact of dose-dense neoadjuvant chemotherapy on pathologic response and survival for HER2-positive breast cancer patients who receive trastuzumab | npj Breast Cancer

Trastuzumab in Female Breast Cancer Patients With Reduced Left Ventricular Ejection Fraction | Journal of the American Heart Association