Dual dose-dependent effects of fingolimod in a mouse model of Alzheimer's disease | Scientific Reports

Acute Fingolimod Effects on Baroreflex and Cardiovascular Autonomic Control in Multiple Sclerosis - Vittorio Racca, Marco Rovaris, Rosella Cavarretta, Emanuele Vaini, Anastasia Toccafondi, Marco Di Rienzo, 2019
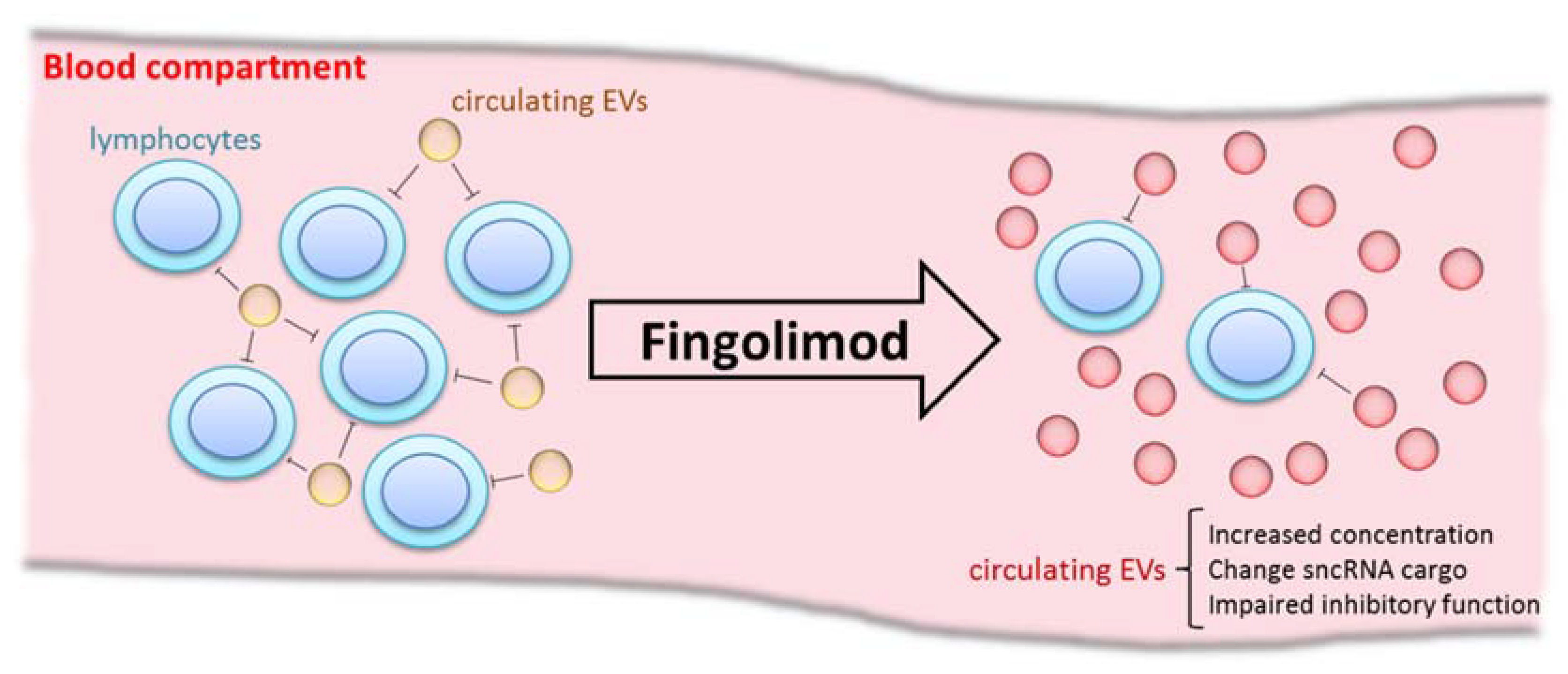
IJMS | Free Full-Text | The First Dose of Fingolimod Affects Circulating Extracellular Vesicles in Multiple Sclerosis Patients
![Teva Canada Announces the Launch of a Bioequivalent Generic Version of [Pr] Gilenya®, [Pr]Teva-Fingolimod Capsules for the treatment of Relapsing-Remitting Multiple Sclerosis (MS) Teva Canada Announces the Launch of a Bioequivalent Generic Version of [Pr] Gilenya®, [Pr]Teva-Fingolimod Capsules for the treatment of Relapsing-Remitting Multiple Sclerosis (MS)](https://mma.prnewswire.com/media/1019389/Teva_Canada_Limited_Teva_Canada_Announces_the_Launch_of_a_Bioequ.jpg?p=facebook)
Teva Canada Announces the Launch of a Bioequivalent Generic Version of [Pr] Gilenya®, [Pr]Teva-Fingolimod Capsules for the treatment of Relapsing-Remitting Multiple Sclerosis (MS)

An Effective Approach to Assess Bioequivalence of Fingolimod in Normal Healthy Volunteers Using 0.5 mg Study Dose