COVID-19 Vaccine Second-Dose Completion and Interval Between First and Second Doses Among Vaccinated Persons — United States, December 14, 2020−February 14, 2021 | MMWR

Coronavirus (COVID-19) Update: FDA Shortens Interval for Booster Dose of Moderna COVID-19 Vaccine to Five Months | FDA

Safety and immunogenicity of one versus two doses of the COVID-19 vaccine BNT162b2 for patients with cancer: interim analysis of a prospective observational study - The Lancet Oncology

COVID-19 Vaccine Second-Dose Completion and Interval Between First and Second Doses Among Vaccinated Persons — United States, December 14, 2020−February 14, 2021 | MMWR
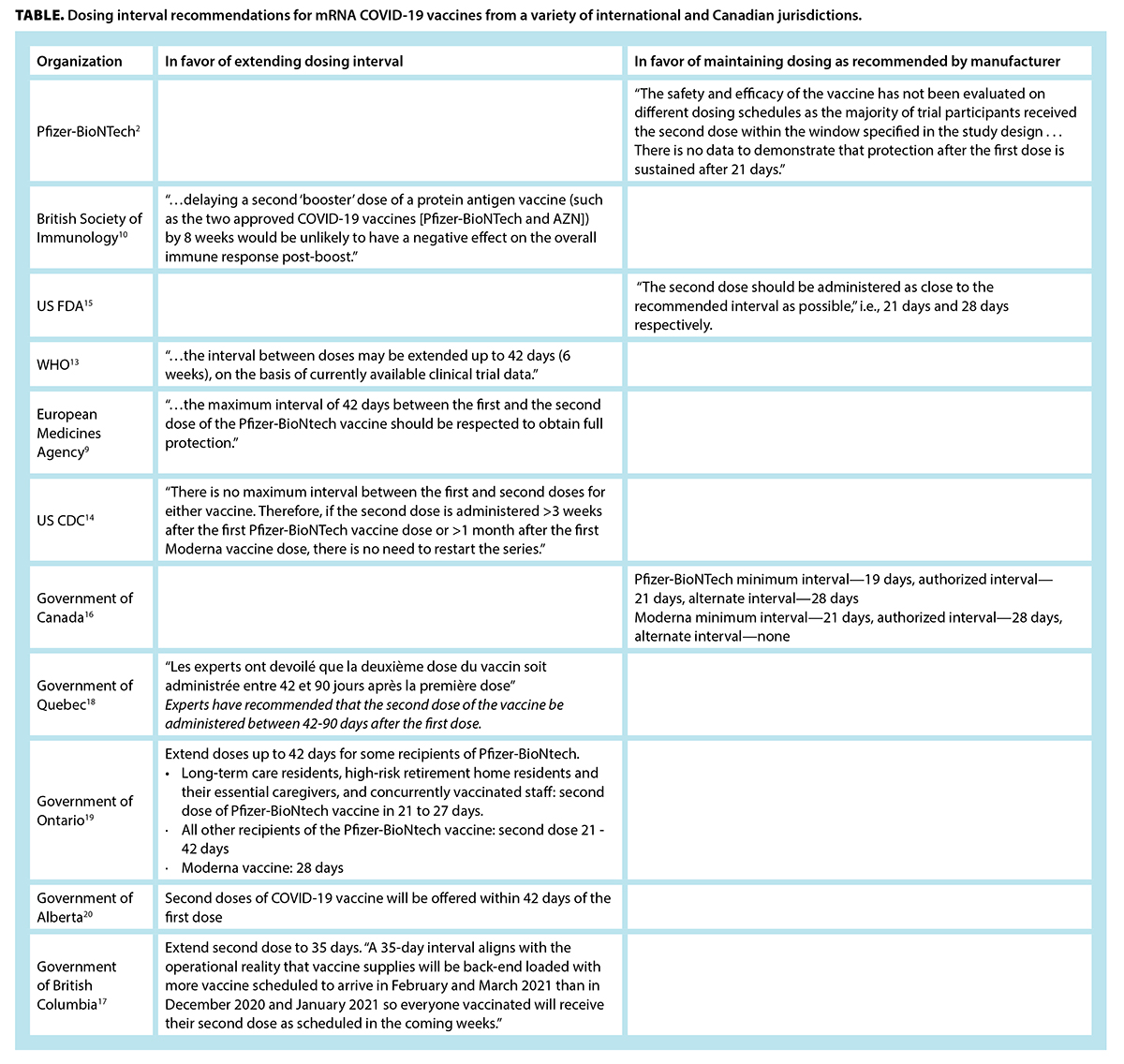
What is the evidence for extending the SARS-CoV-2 (COVID-19) vaccine dosing schedule? | British Columbia Medical Journal

Is there a minimum and maximum time interval between COVID-19 vaccine doses? | COVID-19 Info Vaccines

Effectiveness of the Pfizer-BioNTech and Oxford-AstraZeneca vaccines on covid-19 related symptoms, hospital admissions, and mortality in older adults in England: test negative case-control study | The BMJ


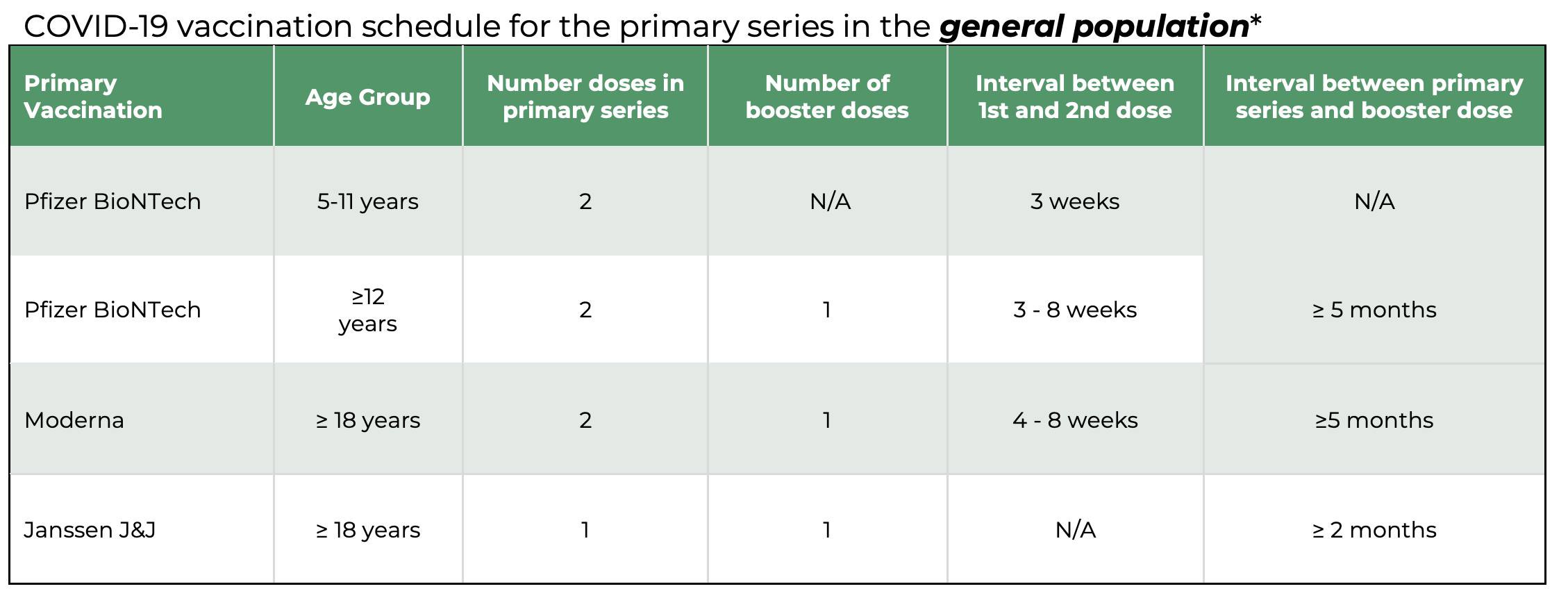

.jpg)